Aniridia Basics
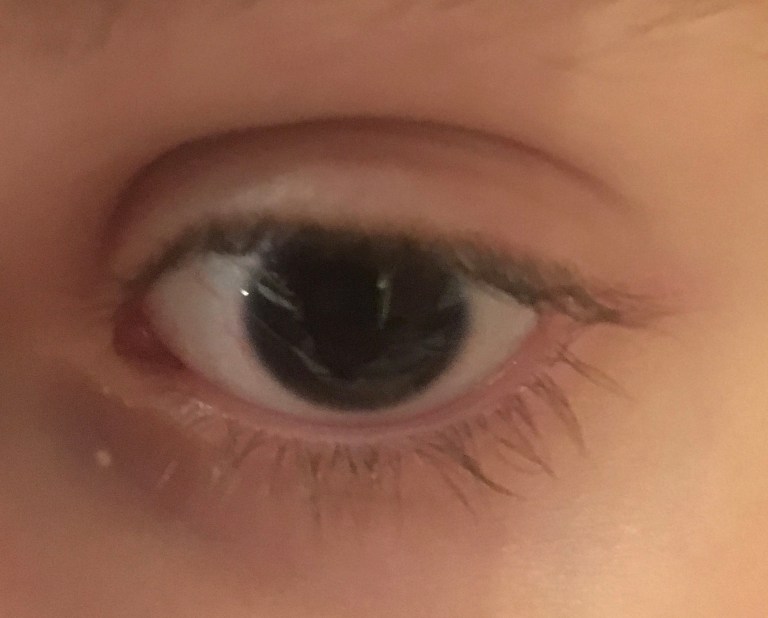
Aniridia is a rare genetic condition generally characterized by either the complete or partial absence of the iris, which is the colored part of the eye.
The word “aniridia” means “without iris”. The lack of iris is often the first and most noticeable feature of aniridia. Typically, a parent or a pediatrician will notice that the child has large pupils, that the child’s pupils do not react to light, or that the child has black eyes with no discernible color.
Although the name “aniridia” focuses on the iris, the condition is panocular, which means that it can affect the entire eye. Complications can occur in other portions of the eye anatomy, including the cornea, lens, anterior chamber, retina, tear film, and optic nerve.
Recent research indicates that non-ocular medical conditions can also occur with aniridia, including sleep disturbances, auditory processing disorder, and issues with pancreatic functioning.
Visual Acuity
Aniridia affects vision differently for each individual. Most people with aniridia are considered to be visually impaired, which means that their vision cannot be fully corrected with standard glasses or contact lenses.
The level of visual impairment depends on many factors, including the number and degree of associated eye complications that a person experiences over time. Some people with aniridia are legally blind, while others have enough vision to drive a car.
Best Corrected Visual Acuity (BCVA) is a measure of a person’s best possible vision when corrected with glasses, as determined by an ophthalmologist using a Snellen eye chart. The BCVA is represented by a fraction – the type of fraction used depends on your country of origin. 20/20 or 6/6 vision is considered normal vision, whereas 20/200 or 6/60 vision is considered legally blind in most countries.
Aniridia Genetics
Genes are the building blocks of all life, as they hold the instructions to build and maintain an individual’s cells. Any changes in a person’s genes can have significant consequences.
Aniridia is caused by either a genetic mutation or deletion. This mutation or deletion causes the gene to function incorrectly.
The vast majority of cases of aniridia are caused by a mutation or deletion in the PAX6 gene, which is located on the short arm of the 11th chromosome. Occasionally, aniridia is caused by mutations in the genes PITX2, FOXC1, and others.
Aniridia affects between 1:40,000 and 1:100,000 people and seems to affect genders and races equally.
Two-thirds of cases of aniridia are inherited in an autosomal dominant manner, which is called “familial aniridia”. One-third of cases are a spontaneous mutation, which is referred to as “sporadic aniridia”.
Some cases of sporadic aniridia are caused by a larger deletion encompassing the PAX6 gene, the WT1 gene, and other nearby genes. These larger deletions result in WAGR Syndrome.
In a very small percentage of cases, aniridia can be inherited in an autosomal recessive pattern. This is called Gillespie Syndrome and is associated with cerebellar ataxia and intellectual disability. Due to its rarity, the genetic cause of Gillespie Syndrome is unclear.


WAGR Spectrum
Aniridia may occur as a part of WAGR Spectrum. When this occurs, there may be additional medical complications. “WAGR” is an acronym for the most common features:
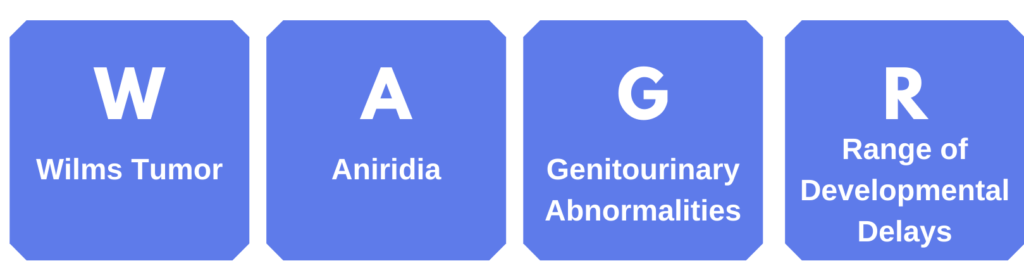
WAGR Spectrum results from a deletion of more than one gene on the 11th chromosome. Typically, this includes the PAX6 gene, the WT1 gene, and surrounding genes; however, the specific genes that are deleted varies between individuals.
Treatments for Aniridia
Treatments for Aniridia focus on preserving vision, maximizing the use of vision, and learning compensatory strategies for living full lives regardless of vision.
Regular visits with ophthalmologists are required in order to monitor and treat any associated eye complications. Drug therapies can be of use for some conditions. Surgical interventions are also sometimes required; however, a conservative approach to surgery is very important in an aniridic eye.
Low vision optometrists, teachers of the visually impaired, and orientation and mobility specialists work with people with aniridia to determine the proper tools to maximize the use of vision and to teach compensatory skills. Magnifiers, monoculars, CCTVs, tablets, and other tools can be used to compensate for visual impairments.
Associated Eye Conditions
Glaucoma
Glaucoma is the result of increased pressure within the eye, which, if not treated, can permanently damage the optic nerve and cause vision loss. Juvenile onset glaucoma is frequent in aniridia patients, whereas it tends to occur in older people in the general population. Therefore, testing for glaucoma should begin at birth.
Cataracts
Cataracts are areas of clouding in the lens of the eye and occur in 50-85% of aniridic patients. Juvenile cataracts are common in aniridia patients but should only be removed surgically if they interfere with usable vision.
Strabismus
Strabismus is a misalignment of the eyes, where an eye “wanders” when looking straight at an object. This can be corrected if addressed early with either surgery or patching.
Amblyopia
Amblyopia, also called “lazy eye”, occurs when the brain does not fully acknowledge the vision being sent to it by one eye. Anything that interferes with clear vision in the eye during the first 6 years of life can create an amblyopic eye, including strabismus, ptosis, or cataract. Treatment early in life is important to strengthen vision.
Ptosis
Ptosis is an abnormal drooping of the upper eyelid. If the ptosis is severe enough to block vision, it can be treated with surgery.
Dry Eye
Poor quality tears are associated with aniridia, which can cause dry eyes. Dry eyes can, in turn, aggravate the corneal problems observed in Aniridia. Preservative-free artificial tears can help to keep the eye moist, as can autologous serum eye drops.
Corneal Keratopathy
In a normal eye, the cornea is a clear covering over the pupil and iris. In up to 90% of aniridic patients, corneal abnormalities such as clouding and an overgrowth of blood vessels can occur. These abnormalities are called aniridia-associated keratopathy (AAK). Although this begins early in life, it typically doesn’t affect vision until adulthood.
Photophobia
Photophobia is increased light sensitivity, due to the lack of iris in the eye. Although indoor lighting is not harmful to someone with aniridia, it is common for some lights to be more comfortable than others. Bright sunlight can often be uncomfortable for people with aniridia, so sunglasses and hats are recommended even for infants and young children.
Nystagmus
Nystagmus is the term for involuntary eye movements. Sometimes referred to as “wobbly eyes”, it is believed that nystagmus is the result of foveal hypoplasia. Nystagmus in aniridia typically presents within the first 2-3 months of life, if not at birth.
Hypoplasia
Hypoplasia means “underdevelopment” of a tissue or organ. People with aniridia frequently have foveal or optic nerve hypoplasia. The fovea is the very center of the retina and is responsible for most detailed vision. The optic nerve carries the messages from the eyes to the brain. Hypoplasia is a frequent cause of baseline visual impairment in aniridia and cannot be corrected with glasses.
Aniridic Fibrosis
Aniridic Fibrosis Syndrome (AFS) is a potential complication resulting from surgery in an aniridic eye. Fibrous tissue forms in the anterior chamber of the eye, causing vision loss. Surgical intervention is often required when AFS is present.
Associated Medical Conditions
Recent research indicates that non-ocular medical conditions can occur with aniridia. The PAX6 gene is a master regulatory gene expresssed in multiple areas of the body, a change in that gene can have systemic effects. The conditions described below have been scientifically linked to aniridia caused by PAX6 mutations.
CAPD
Central Auditory Processing Disorder (CAPD) involves difficulty with processing what a person hears, particularly in a noisy environment. It is believed that CAPD is caused by abnormal interhemispheric transfer in the aniridic brain.
Subtle Brain Abnormalities
Aniridia has been linked to brain abnormalities in the anterior commissure, anterior cingulated cortex, cerebellum, temporal and occipital lobes, corpus callosum, pineal gland, and olfactory bulb.
SPD
Sensory Processing Disorder (SPD) is a condition where the brain struggles with receiving and responding to the messages sent from the senses. This can include being extra sensitive or under sensitive to sensory stimulation.
Sleep Disturbances
Research has shown that people with aniridia often have an underdeveloped pineal gland and lower levels of melatonin. This can create isses with falling asleep and staying asleep.
Altered Olfaction
People with aniridia often have a decreased, altered, or absent sense of smell. Some report a heightened sense of smell. Imaging has shown that the olfactory bulb is often underdeveloped, contributing to this altered olfaction.
Pancreatic Issues
Abnormalities in the structure or function of the pancreas are common in aniridia. This can range from mild glucose intolerance to insulin resistance to diabetes. Research is ongoing to determine how this may be different than typical diabetes.
Additional Resources
Further information about aniridia can be found at the links below:

